
By Jennifer Kuzma*, Goodnight-NC GSK Foundation Distinguished Professor and Co-Director, Genetic Engineering and Society Center
*Note: The opinions expressed in this article are those of the author as an individual faculty member and should not be taken as a reflection of the views of the whole of the Genetic Engineering and Society Center or NC State University.
In recent years, the regulatory system for biotechnology products has not kept pace with newer ways of engineering organisms, such as through the use of gene editing like CRISPR-Cas9 systems (see Kuzma 2016 Nature 531:165).
Under the Obama administration, progress had been made in clarifying U.S. biotechnology regulations. In January 2017, in the last few days of Obama’s term, several proposals were made for updating agency regulations and guidance documents. In particular, new US Department of Agriculture (USDA) regulations were proposed for genetically engineered (GE) crops.
Fast forward ten months to November 6, 2017, and the Trump administration has pulled this proposed rule back to “start fresh” and reconsider the issue. This is no surprise, as it is not uncommon for new political administrations to recall regulatory policy for biotech and other products. Many industry and academic scientists developing GE crops are pleased to hear about the Trump administration’s recall of USDA proposed regulations (see Science: Trump’s agriculture department reverses course on biotech rules).
However, I saw the proposed rule as a thoughtful, balanced approach for dealing with new gene-edited crops, as well as for aligning the regulations to comply with modern agricultural-protection laws. So, although I am not surprised, I am disappointed by the recall of USDA’s proposed Jan 2017 rules. I explain below.
Science: Trump’s agriculture department reverses course on biotech rules
The 1986 White House policy document, The Coordinated Framework for the Regulation of Biotechnology (CFRB), argued that as a broad category, genetically engineered products pose no new types of risks when compared to conventionally-bred products, and that regulation should be focused on the product not on the process by which it is produced. Hence, the CFRB suggested that no new laws were needed to regulate the products of biotech and existing laws should be used.
As a result, the USDA began regulating GE crops in the 1990s under the 1957 Federal Plant Pest Act. At the time, the “hook” to capture GE crops was that they contained DNA sequences from plant pests.
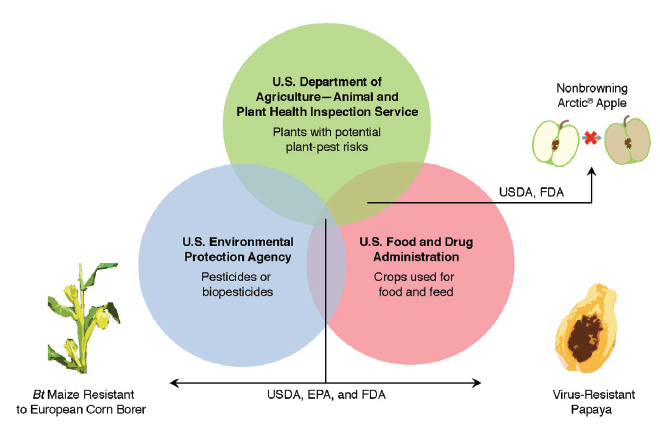
U.S. regulatory agencies that have responsibility for genetically engineered (GE) crops. NOTE: Depending on the GE trait in question, evaluation by one or all three of the agencies within the Coordinated Framework may be required before commercial release of a GE crop. For example, GE virus-resistant papaya went through the regulatory process of all three agencies. The use of Agrobacterium tumefaciens to transfer virus resistance was classified as use of a plant pest by APHIS, EPA classified virus resistance as conferring pesticidal quality, and consultation was completed with FDA because the papaya was intended for human consumption. In contrast, GE non-browning apple required evaluation by only two agencies because A. tumefaciens was used (APHIS) and food-safety assessments were required (FDA). Evaluation by EPA was not required because the gene responsible for the non-browning trait was not classified as a plant-incorporated protectant. Source: National Academies of Sciences, Engineering, and Medicine. 2016. Genetically Engineered Crops: Experiences and Prospects. Washington, DC: The National Academies Press. doi: 10.17226/23395 (Chaired by Fred Gould, Co-Director of GES Center)
The first generation of GE crops was made possible by engineering sequences from Agrobacterium alongside the genes of interest so that the DNA would insert itself into the genome of the host plant. Sequences from another plant pest, the Cauliflower Mosaic Virus (CaMV) were used to keep the genes of interest turned on, or expressed, in the host plant. Because the GE crops had plant pest sequences, USDA could capture them under the Federal Plant Pest Act of 1957. Although this shaky basis for regulation concerned some scientists and NGOs, the result was that USDA regulated all GE crops by requiring field trial permits or notifications, and deregulation petitions and environmental assessments. (See National Academy of Sciences 2000 Genetically Modified Pest Protected Plants report.)
However, in the new millennium this changed as new genetic engineering technologies were used to get the DNA into the host plant (called “transformation”) and to make the intended genetic change. GE crop developers no longer had to rely on plant-pest DNA sequences.
USDA had a choice to make—should its key biotech regulatory agency, APHIS, capture new GE plants made these new ways, or not?
The issues with gene-edited crops and engineered crops resulting from new transformation methods started to come to light in popular and academic press (see for example Waltz 2011 Nature Biotech 29: 772 and Kuzma & Kokotovich 2011 EMBO Reports 12:883). It was discovered that USDA had been exempting GE crops like herbicide tolerant grass or reduced phytate gene-edited corn (made possible without plant pest sequences) without any public or federal advisory committee discussion.
Instead, letters between producers and regulators – in which USDA decided that the new GE crops did not fall under their plant pest regulations – were made public only after Freedom of Information Act (FOIA) requests (Waltz 2012 Nature Biotech 30:215).
Since then dozens of GE crops have undergone no regulatory assessment prior to market. And now, they are coming to market.
In 2015, as more groups became concerned about the exemption of most new GE crops, and the issues were made public in the media, the White House under the Obama administration asked key federal agencies to reconsider and update biotech regulations under the CFRB. In September 2016, after a year of public and stakeholder consultation, they finished the first phase of that process (Fed Reg 81: 65414).
Although I criticized it for its limited scope (it merely clarified existing regulations, without revising them) and insufficient public engagement (see Kuzma 2016 Science 353:1211) I viewed it as a step in the right direction in bringing regulations up-to-date with emerging technologies.
Soon after, a more specific proposal to make substantive changes to USDA’s regulations was released during the last week of the Obama administration (January 19, 2017). USDA re-wrote its regulations to bring them up to speed with the most recent plant protection law.
In particular, in 2000, the Plant Protection Act replaced the Federal Plant Pest Act of 1957, by combining both the plant pest and noxious weed regulatory authorities in USDA. In USDA’s January 2017 proposed rule, GE crop regulations were now designed to conform to the new law and promised to include a look at potential weed risks related to GE crops, in addition to continuing to look at plant pest risk.
Weed risk issues have been of greatest concern from the planting of first generation GE crops.
In recent years, there has been a marked increase in weeds resistant to Roundup – caused by overuse of the herbicide in conjunction with Roundup tolerant (‘Roundup Ready’) GE crops that were engineered to be resistant to the herbicide.
Weed risks resulting from the planting of first generation GE crops have been demonstrated, and USDA’s proposal to use its noxious weed authorities to assess GE crops made scientific sense to me. Additionally, this would also put the agency in legal compliance with the most recent law on plant protection.
USDA also proposed in the new 2017 regulations that it would analyze a greater number of GE crops that were generated from gene editing. Again, they had been exempting, behind closed doors, the vast majority of gene edited crops from their regulatory review and risk assessment process (e.g. because these gene-edited crops were not engineered with sequences from plant pests). Now the agency would review many gene edited crops (except those with single point mutations that could be produced through conventional mutagenesis) and assess them for both plant pest and weed risk potential – two science-based characteristics that associate with risks to agriculture.
If there seemed to be a significant concern from the analysis step, USDA would require field trials and more data generation under the new regulations. It was an “analyze first, regulate second” policy that brought more crops under the initial umbrella of assessment as compared to the old rules.
This also made great sense to me scientifically and practically – to take a look first, base an assessment on risk factors for weediness and pest-like characteristics, and then decide whether to fully regulate. It was risk-based and acknowledged the practical difficulties of not being able to (or wanting to) regulate everything or nothing (Kuzma 2016 Nature 531:165).
Scientists with conflicts of interest have raised concerns.
It is confusing now that some scientists developing GE crops are citing a lack of “scientific basis” in the proposed rules, and in my view, this is really just a cover for arguing against any more regulation. Impeding innovation was really at the heart of the matter.
These days, most plant biotechnologists know that the trick to avoid regulation is to avoid the use of plant pest DNA sequences. This is not a scientific basis for regulation, as these partial DNA sequences do not on their own act as “plant pests.” As a result, our immediate future will be that most new GE crops will be engineered without these sequences, and thus, most will undergo no USDA regulation and potentially no regulation at all if EPA pesticide authorities are not triggered.
What prompted this pullback?
Federal agencies are influenced by Congress, as it controls their budgets. In October 2017, Congressional committees in the House raised concerns about U.S. biotech rules confusing trading partners.
I suspect that this prompted the Secretary of USDA to pressure the career-agency USDA scientists to withdraw the rule on November 6, 2017.
It really has little to do with USDA’s stated rationale, which is two-fold:
- In the public comments there were objections from both sides – the more pro-technology as well as the more precautious sides – on the same points, meaning that the rule was somewhat flawed and therefore,
- USDA is legally obligated to withdraw it in order to consider other alternatives in public settings.
The agency then suggests it will “start fresh” and consult with a broad range of stakeholders to consider more diverse alternatives.
However, I don’t think that is really what is going on.
My thinking is that if a rule is criticized by both sides on the same points (in this case, what is exempted AND what is to be included in the initial analysis step), it has probably struck a good balance.
USDA also engaged stakeholders through public meetings and comments already on the draft environmental impact statement and proposed rule in 2016 and up to summer 2017.
Instead, what is going on is that politics and worldviews motivate decisions.
The underlying reason for the withdrawal is based on concerns that more crops would be captured for analysis by a regulatory agency, and this new administration isn’t too fond of regulations in general (an understatement perhaps…).
Regulations always have a value component to them and can’t be based strictly only on scientific risk (see Kuzma Nature), so the new administration is exerting its values which are more anti-regulatory. These values match the pro-technology, less precautious interest groups that generally have more power (e.g. industry and biotech developers). It just took a while for the political appointees in USDA to clue into what was happening at the career agency-scientist level and stop it.
So again, this wasn’t unexpected, but I am surprised that it would pull the rule so late after the start of the Trump administration, allowing USDA staff to expend its precious resources by hosting public meetings up until July 2017 to consider the new rule. Perhaps it just took more time for this new administration to get in place and figure out what was going on (again, another understatement…)
I personally think the new rule was a good compromise position between the regulate nothing/everything divide that occurs from a strict “science-based” perspective (see Kuzma Nature 2016). It made scientific sense to include the noxious weed authorities as these risk issues have been of greatest concern for first generation GE crops, and it made scientific sense (though perhaps not by strict quantitative risk) to exclude single point mutations and those otherwise that could have happened through conventional breeding. It also makes scientific sense to analyze gene-edited and other crops made without plant pest sequences for plant pest and weediness risk.
So, I am quite disappointed by all the effort wasted in the formulation of the new rules, and the revisiting of the CFRB generally. The proposed rule might have some scientific and practical flaws, but it was surely more scientific than our current regulations, despite what those with conflicts of interest (prominent molecular biologists) who want to maintain the status quo of no regulation for many GE crops might say (“in the name of science…”). In fact, these scientists are now reversing their 20+ year-old arguments that the process of engineering should not matter by wanting USDA to stick with process based regulatory triggers. This is hypocritical (Kuzma Nature 2016)
Politics “trumps” science again, and this time the “scientific mainstream” dominated by conflicted biotech developers is on board.
About
Dr. Jennifer Kuzma is the Goodnight-NCGSK Foundation Distinguished Professor, School of Public and International Affairs, and Co-Director, Genetic Engineering and Society Center, at NC State University. Kuzma is a social and policy scientist who has spent time in the late 1990s-early 2000s in practical policy positions in Washington DC as a AAAS fellow at USDA Office of Risk Assessment and Cost Benefit Analysis and as a study director at the National Academy of Sciences.
Since returning to academe in 2003, Kuzma has served on several national and international advisory boards, including the World Economic Forum’s Global Futures Council on Technology, Values, and Policy and the U.S. National Academy of Sciences Committee on Preparing for Future Biotechnology. She has over 100 scholarly publications on emerging technologies and governance; and has been studying biotechnology and its societal aspects for over 25 years. She obtained a Ph.D. in plant biochemistry from the University of Colorado Boulder in 1995 and completed a post-doc in Plant Molecular Biology at the Rockefeller University 1995-1997 before entering the world of policy sciences.